Why the Withdrawal Was Ordered
The European Medicines Agency (EMA) has decided to withdraw all medicines containing levamisole from the European market.
This decision follows a safety review by the Pharmacovigilance Risk Assessment Committee (PRAC).
After analyzing available data, experts concluded:
The risks now outweigh the benefits
European law allows withdrawal when a drug’s safety profile becomes unfavorable
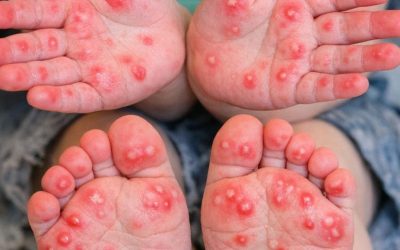
Main Concern: Risk of Leukoencephalopathy
The key issue is the risk of Leukoencephalopathy, a condition affecting the brain’s white matter.
Why this matters:
White matter enables communication between brain regions
Damage can impair neurological function
According to EMA findings:
The condition is rare but serious
It can occur even after a single dose
Symptoms may appear:
Within 24 hours
Or months after treatment
What Was Levamisole Used For?